(openPR) (Amherst, NH) – The evolution in drug injection devices to accommodate new injectable drugs designed for self-administration continues to change the therapeutic drug landscape. Attempting to address key healthcare issues that include patient safety and drug protocol compliance, device designers are now building on the experience of earlier generations of syringes and injectors, incorporating new design features with the aim of making the injection event more consistent across patient populations and less likely to evoke feelings of apprehension.
In this environment, pen injectors have found strong user acceptance for a number of chronic conditions, a list that includes autoimmunity, diabetes, osteoporosis and reproductive medicine. On a global basis, the growth of pen injectors in these therapeutic segments continues to climb, already representing the majority of injections in several, including hormone replacement and selected metabolic diseases (diabetes, osteoporosis).
While pen injectors represent a significant cost premium over syringes, drug companies are finding that customized pen devices can be important product differentiators in direct-to-consumer markets. This awareness has created opportunities for specialty pen device suppliers such as Ypsomed, which continues to dominate in this market. As pen devices become more entrenched in the strategies of injectable drug developers, alternative device acquisition strategies will put pressure on the traditional outsourcing models.
Detailed analysis of pen injector products, therapeutic sectors, and participants is included in a new and comprehensive report. The report documents and analyzes the results of a recent survey of pen injector drug delivery conducted by Greystone Research Associates.
More information is available at
http://www.greystoneassociates.org/Pen_Injectors.htm
Source: Greystone Research Associates
Presseinformation
Pen Injectors Becoming Commonplace in Self-Administration for Key Chronic Indications
Diese Pressemeldung wurde auf openPR veröffentlicht.
Verantwortlich für diese Pressemeldung:Contact:
Mark Smith
Greystone Research Associates
98 Route 101A
Amherst, NH 03031 USA
Voice: 603-595-4340
Fax: 603-218-7020
www.greystoneassociates.org
Mark Smith
Greystone Research Associates
98 Route 101A
Amherst, NH 03031 USA
Voice: 603-595-4340
Fax: 603-218-7020
www.greystoneassociates.org
Über das Unternehmen
About Greystone
Greystone Associates is a medical technology consulting firm focused on the areas of medical market strategy, product commercialization, venture development, and market research. We assist medical and healthcare market participants in achieving their business objectives through the creation of detailed development strategies, product commercialization programs, and comprehensive market and technology research and analysis. Our market research publications are designed, researched and written to provide timely and insightful information and data on focused market segments, with the aim of providing market participants with the essential knowledge to refine and execute their marketing plans and financial targets.
Greystone Associates is a medical technology consulting firm focused on the areas of medical market strategy, product commercialization, venture development, and market research. We assist medical and healthcare market participants in achieving their business objectives through the creation of detailed development strategies, product commercialization programs, and comprehensive market and technology research and analysis. Our market research publications are designed, researched and written to provide timely and insightful information and data on focused market segments, with the aim of providing market participants with the essential knowledge to refine and execute their marketing plans and financial targets.
Pressebericht „Pen Injectors Becoming Commonplace in Self-Administration for Key Chronic Indications“ bearbeiten oder mit dem "Super-PR-Sparpaket" stark hervorheben, zielgerichtet an Journalisten & Top50 Online-Portale verbreiten:
Disclaimer: Für den obigen Pressetext inkl. etwaiger Bilder/ Videos ist ausschließlich der im Text angegebene Kontakt verantwortlich. Der Webseitenanbieter distanziert sich ausdrücklich von den Inhalten Dritter und macht sich diese nicht zu eigen. Wenn Sie die obigen Informationen redaktionell nutzen möchten, so wenden Sie sich bitte an den obigen Pressekontakt. Bei einer Veröffentlichung bitten wir um ein Belegexemplar oder Quellenennung der URL.
Weitere Mitteilungen von Greystone Research Associates

Prefilled Syringes to Reach 4.75 Billion Units in 2016 According to Greystone Research
(Amherst, NH) – Driven by improved device designs, a maturing supply chain infrastructure and broad acceptance of inherent patient and caregiver benefits, prefilled syringes will continue to grow at a double digit rate, climbing to four and three quarter billion units worldwide in 2016, according to the latest estimates from Greystone Research Associates.
Drug developers seeking to package their injectable formulations in prefilled devices need an understanding of the unique technology and business factors defining this sector. The prefille…

Growth of Specialty Injectable Drugs Placing Emphasis on Device Evolution and User Perceptions
(Amherst, NH) – As prefilled injection devices continue to replace vials in terms of injections as well as total sector revenue, pharmaceutical and biotech program managers are increasingly integrating syringe and self-injection device technology into their development plans. The result has been a migration toward multi-functional development teams and an increasing reliance on accelerated product feasibility testing. As the factors driving this trend continue to evolve over the next four years, the number of tailored injectable drug products…
Das könnte Sie auch interessieren:

Innovative Personalentwicklung mit AGON-PEN
… Kommunikationswegen, den aufwändigen, zeitraubenden und kostenintensiven Papierproduktions- und -bewegungsmechanismen ist dazu fast immer zu schwerfällig.
Die neue "Personalentwicklung mit AGON-PEN ermöglicht es dem Unternehmen in einem Performance-Management-Prozess einen dezentralen Personalentwicklungsprozess mit zentraler Kontrolle und Evaluation der Ergebnisse zu …


Eisbach Partners: Robert Maier as new Associate Partner
Digitization and new technologies are shaking up markets, innovation cycles are becoming shorter, companies are competing worldwide. This underpins the necessity to not only identify new business models quickly and effectively but also the urge to have a fast and goal-oriented implementation. Therein, the customer is key - Only if the customer’s desires …

Dry Powder Inhalers Continue to Dominate Pipeline Development for Inhaled Systemic Therapeutics
… preserve their shelf life and require continuous refrigeration.
As aging population demographics and managed care initiatives drive growth in home health care and self-administration of drug therapies, inhaled medicine is increasingly being viewed as patient-friendly and cost-effective. Our analysis demonstrates that inhaled administration in general, …
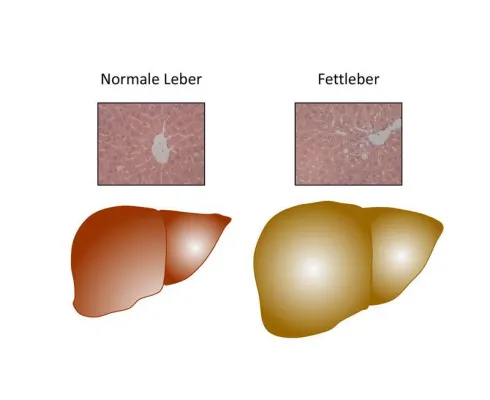

Trigger for Fatty Liver in Obesity
… can lead to liver steatosis and insulin resistance, even in case of a normal body weight,” Konrad explains.
Permeable mitochondria inhibit lipid oxidation
The team of researchers found indications that an activation of Fas affects the mitochondria: Their capacity to oxidize fatty acids is limited and therefore leads to the accumulation of lipids in liver …


On the way to developing a new active ingredient against chronic infections
… keep both patient groups in mind when improving the structure of their lead compound. As proven in various test systems, the starting molecule has good conditions for becoming a successful active ingredient: The pathoblockers inhibit the function of the PqsR receptor, which plays a key role in the infection of Pseudomonas aeruginosa. The bacterium uses …

Growth of Specialty Injectable Drugs Placing Emphasis on Device Evolution and User Perceptions
… growth hormone (hGH). A new class of devices – the prefillable safety syringe – is positioned to make serious inroads into both the clinical and self-administration healthcare segments.
Detailed analysis of Specialty Injectable Drugs, therapeutic sectors, and participants is included in a new and comprehensive report. The report documents and analyzes …

Dual Chamber Devices for Drug Reconstitution Grow in Response to Ease-of-Use Needs of Self-Medicating Patients
… increasing interest in injection devices that can eliminate the number of steps required to reconstitute the drug and make it available for injection. Dual chamber syringes and injectors – devices that allow reconstitution to take place within the device immediately prior to injection - are the most elegant engineering solution to this need.
The current …


Cerbomed at the INS 10th World Congress
… two poster presentations.
About NEMOS
NEMOS is intended for people suffering from various difficult to treat neurological and psychiatric diseases. The current lead indications are drug-resistant epilepsies. Further indications such as chronic pain, tinnitus and schizophrenia are currently being evaluated in clinical studies.
Cerbomed’s t-VNS therapy …

CT-PEN sorgt mit digital Pen & Paper bei Kapsch BusinessCom AG für schnelle Erfassung von Serviceleistungen
… einfacher und günstiger geschehen kann, führte die Kapsch BusinessCom AG den Einsatz digitaler Stifte von Anoto ein. Umgesetzt wurde das Projekt vom Spezialisten CT-PEN Digital Pen & Paper in Salzburg.
Aus Wochen und Tagen werden Minuten
Zur Protokollierung der verrichteten Arbeiten nutzten die Servicetechniker bisher herkömmliche, von Hand auszufüllende …


Schliessanlagen, Check-in Automation & Zutrittsmanagement von Key&Card AG Einfach. Genial. Digital.
Im Gespräch mit Helmut Koch, Geschäftsführer der Key&Card AG, entdecken wir die faszinierende Welt der einfach genialen digitalen Lösungen für Schließanlagen, Check-in Automation und Zutrittsmanagement für die Hotelindustrie.Frage 1: Wie verbessern die Schließanlagen von Key&Card AG die Sicherheit und den Komfort für Hotels?Helmut Koch: Unsere …
Sie lesen gerade: Pen Injectors Becoming Commonplace in Self-Administration for Key Chronic Indications